
Bijay Bahadur
has more than 30 years of experience in Indian/multinational breweries, IMFL bottling plant and brewery green field and brown field projects and factory operations. Author of Brewing – A Practical Approach, is for practicing brewers, students pursuing careers in Brewing Science and consultants who provide technical advice to the breweries and entrepreneurs. He is a FELLOW of The Institute of Engineers (India) and Chartered Engineer (India), lifetime member of the Indian Institute of Chemical Engineers and Association of Food Scientist & Technologist (India). He is also selected as Professional Engineer by ECI, New Delhi. Mr. Bijay is currently associated with Yuksom Breweries Ltd (Sikkim) as Factory Manager (Unit Head).
Beer is a universally popular beverage, consumed worldwide. No one can deny that beer industry has changed drastically over the last three decades both in consumer preferences and production methods.
In addition to increased competitions, there have also been changes in legislation, the lowering of trade barriers and new processing techniques that breweries need to make themselves familiar with in order to remain as competitive as possible.
In fact, in some countries low alcohol beer have been the fastest growing sectors of the alcoholic beverages market. The reason for this is simple. People enjoy having a beer with lunch, but don’t want that sleepy feeling when they return to work. More importantly they want something that tastes like full strength beer and don’t just look like it.
There are several reasons for this:
- General health concern
- Religious reasons
https://www.brewer-world.com/wp-admin/post.php?post=88168&action=edit# - Excise duty
- Stricter drink driving laws
- Although reduced alcohol beer was not considered as “real beer” in the past, techniques for their production have changed, allowing low alcohol beers to finally be considered as serious alternatives to their full-strength counterparts.
Nowadays, it has been observed that no-alcohol (alcohol-free) and low-alcohol beer is on the rise due to several reasons. What will be the impact of the increasing popularity of these products on the consumption of alcoholic beverages – a big question? There is concern that breweries with no-alcohol (alcohol-free) products are less restricted by brand advertising. More advertising for the no-alcohol and low-alcohol brands will be created. This category is very attractive from a margin perspective owing to both its higher than average revenue and lower excise.
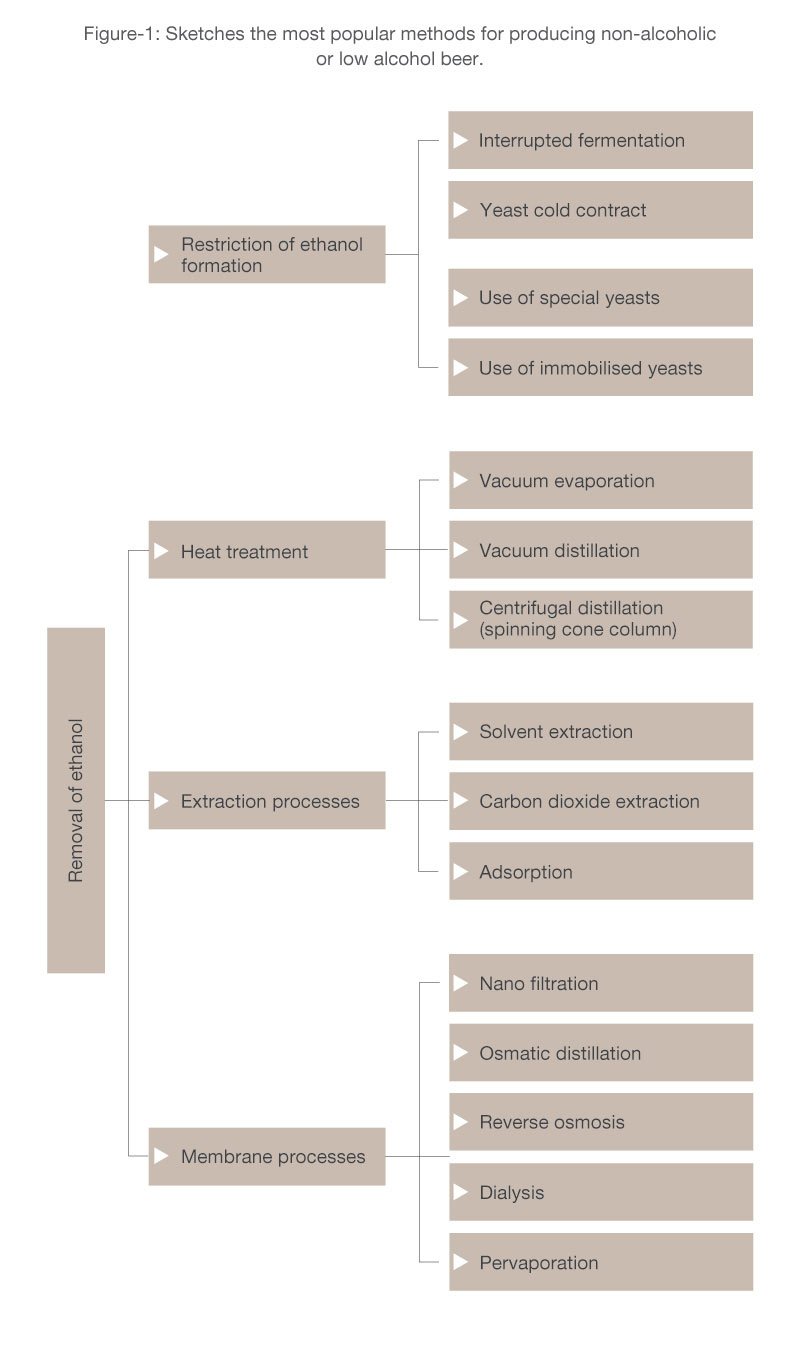
Production of non-alcoholic beers
Usually, the processes for producing non-alcoholic beer can be divided in two major categories:
- Processes of ethanol formation restriction
- Processes of ethanol removal
Restriction of Ethanol Formation Processes
The production of non-alcoholic or low alcohol beer by restricting the ethanol formation can be accomplished by interrupting the beer fermentation and by using special or immobilized yeasts in order to keep the ethanol content very low.
Interrupted fermentation is performed at a temperature lower than a regular fermentation, until reaching an ethanol content below 0.5 % v/v. The fermentation is then stopped by removing the yeast (by centrifugation or filtration) or by flash pasteurization. The beer is then matured at very low temperature (0 – 1oC).
Advantages:
- Uses the same equipment of the standard fermentation.
Disadvantages:
- The formation of aroma compound is restricted
- The final beer is worty aroma
- In yeast cold contact processes, beer yeast contacts with wort at low temperatures, avoiding the ethanol production.
Advantages:
- Uses the same equipment of the standard fermentation.
- Yeast reduces carbonyl compounds from the wort and produces aroma compounds
Disadvantages:
- Yeasts can convert amino acids into aldehydes, which contributes to off flavours
The restriction of ethanol formation can be achieved by substituting typical beer yeasts by special yeast strains, such as Saccharomyces ludwigii species, that ferment fructose and glucose and do not convert maltose, resulting in a beer with low alcohol content (less than 0.5 % v/v) and higher content of sugars.
Advantages:
- Uses the same equipment of the standard fermentation.
Disadvantages:
- High content of sugars in the final beer – sweet taste
- The use of immobilized yeasts is another technology for producing alcohol free beer by avoiding the ethanol formation. The immobilization of yeasts consists of their attachment to a carrier/support, in order to allow the formation of yeast colonies on it. Usually, there are used porous glass particles or brewing and agricultural by-products, such as spent grains or corncobs, as carrier materials. Wort flows continuously through the packed bed of the colonized carrier. The control of operating conditions, such as temperature and wort flowrate (residence time) influences the degree of fermentation, and thus, the alcohol content of the beer at the bioreactor outlet.
Advantages:
- Yeast reduces aldehyde from the wort
- Yeast forms beer aroma compounds
- Good utilization of raw materials
Disadvantages:
- Difficult to control the process
- High price of the carriers
- Risk of contaminations
- Need of continuous bioreactor
Ethanol Removal Processes
In order to obtain a non-alcoholic or a low alcohol beer, ethanol can be removed from a completely fermented beer, and then containing the original aroma profile.
In heat treatment processes, ethanol is removed from the original beer by means of heating. At atmospheric pressure, the ethanol boiling point is 78o C while water boils at 100o C, so applying a temperature lesser than 100o C, alcohol evaporates preferentially and its separation from beer is possible – single or multiple stage distillation. However, high temperature is unbeneficial to heat sensitive compounds, which can suffer physical or chemical modifications. These changes lead to a decrease of beer quality during the dealcoholization process. In order to overcome this disadvantage, low pressure or vacuum can be applied and consequently the separation of ethanol can be achieved at moderate temperatures (lower than 78o C).
In vacuum evaporation beer is heated with steam, under vacuum, in plate evaporators. The evaporated beer flows to a separator where the dealcoholized beer is collected from the bottom and the ethanol rich vapour is led to a condenser. In order to reduce ethanol content even more, alcohol free beer can be made recirculating through the plate evaporator or through multistage evaporators.
Advantages:
- Allows ethanol content of 0.05 % v/v
- Moderate temperatures resulted from vacuum
Disadvantages:
- Need of additional evaporator
- High energy costs
- Thermal impact of sensitive compounds
- Volatile aroma compounds are removed with ethanol
- Vacuum distillation uses typical distillation columns, working under vacuum, for removing ethanol from beer. The bottom product of the distillation column consists of alcohol free beer while the distillate consists of an ethanol rich stream, which is condensed at the distillation column outlet.
Advantages:
- Allows ethanol content of 0.05 % v/v.
- Moderate temperatures, resulted from vacuum
Disadvantages:
- Need of additional distillation column.
- High energy costs.
- Thermal impact of sensitive compounds.
- Volatile aroma compounds are removed with ethanol
- Centrifugal distillation is a variation of vacuum distillation, which uses a column with a special design – the spinning cone column. Spinning cone column (SCC) distillation is a worldwide popular method for beverages dealcoholization. This technology consists in a gas (steam)-liquid counter current column, where the stripping medium (steam) extracts the ethanol from the beverage.
The column comprises a rotating central shaft and spinning cones attached to the shaft inserted with static cones fixed to the column wall. The beer travels down through the stationary cones due to gravity force and flows up through the spinning cones as a thin layer under centrifugal force. The stripping medium (e.g., steam) is fed at the column bottom and collects the alcohol. The vapour with alcohol is carried out of the column for being condensed. The dealcoholized beer flows down the column and is collected from the bottom outlet.
Advantages:
- Allows ethanol content of 0.05 % v/v
- Thermal impact is minimized
- Low residence time and moderate temperatures
- High turbulent flows and high contact area between two phases
Disadvantages:
- Need of additional spinning cone column
- High energy costs
- Stripping medium also removes volatile aroma compounds
- Generally, all heat treatments affect the aroma flavour and taste of the non-alcoholic beer. The applied temperatures (even moderate ones) or the strip streams spoil the beer quality; several important beer compounds are removed and some of beer compounds suffer chemical reactions during heat processing.
Extraction Processes
Extraction processes use an extraction medium to remove ethanol from beer.
In solvent extraction, feed beer contacts with an immiscible phase (the solvent, e.g., pentane or hexane) that presents a good solubility to ethanol. Ethanol is thereby removed from the beer into the solvent.
Advantages:
- Solvents are immiscible with water and present high solubility of ethanol
Disadvantages:
- Additional liquid-liquid extraction unit is needed
- Aroma compounds that are soluble in the solvent are removed
- Product can show traces of the solvent
- Solvents must be food grade and approved by the regulatory body of the Govt.
- Carbon dioxide extraction is based on the same principle of the extraction with solvent (liquid carbon dioxide). Carbon dioxide has properties similar to organic solvents at certain temperature and pressure. This process produces a good quality beverage.
Advantages:
- Carbon dioxide extract ethanol, without extracting water and other larger molecules (proteins and carbohydrates) of beer
- Can be performed at room temperature, avoiding thermal impact of beer
Disadvantages:
- Additional extraction unit is required
- Carbon dioxide strips other volatile compounds
- High operation costs
- Adsorption can be used for beer dealcoholization. This separation process is based on the adsorption and molecular sieving of ethanol in hydrophobic adsorbents, such as zeolites. The original beer is thereby separated in an aqueous stream (alcohol free beer) and an alcoholic adsorbed phase. After ethanol removal, a gaseous stream, such as carbon dioxide, thermally regenerates the adsorbent. Usual adsorbents also extract other beer aroma compounds; thus, an additional distillation column is required for dealcoholizing the desorbed phase, resulting in an alcoholic distillate and an aroma concentrated stream. The last one
is added to the alcohol-free beer for balancing the aroma profile. This approach is very expensive and is only feasible for very specific applications.
Advantages:
- Hydrophobic adsorbents such as zeolites have good affinity with ethanol
Disadvantages:
- Additional separation unit is needed.
- Adsorbents must be regenerated.
- Aroma compounds co-adsorb with ethanol
- High operation costs
Membrane-based processes
Membrane is a semi-permeable physical barrier, placed between two bulk phases, which allows the transport of one or more species between these phases. This transport is controlled, among other factors, by membrane nature. In membrane processes, the product which is intended to be treated (the feed stream) flows tangentially to the membrane surface and is separated in two fractions, the permeate that crosses the membrane and the retentate that remains in the feed side. This type of separation is known as cross-flow filtration and it can be applied to the dealcoholization of beer. Various membrane separation processes can be used differing in the physical effects that allow the ethanol separation.
Nanofiltration:
Advantages:
- Beer is treated at low temperature and pressure
- Membranes show high retention to aroma compounds
Disadvantages:
- Need of additional nanofiltration plant
- Need of diafiltration water
Reverse Osmosis (RO):
Advantages:
- Beer is treated at low temperatures
- Some membranes show high retention to aroma compounds
Disadvantages:
- Additional membrane unit is required.
- High pressure is unbeneficial to the beer
- Some membranes can show low retention to aroma compounds
- Need of diafiltration water
- Difficult to achieve ethanol below 0.45 % v/v.
- Osmotic distillation (OD) or evaporative perstraction (EP) is another membrane process used for removing ethanol from beers. OD uses hydrophobic porous membrane contactors to provide the ethanol transfer from
the alcoholic beverage to the strip solution (usually water) that flows counter-currently at the downstream side of the module.
The mass transport is driven by the difference of the vapour pressure of ethanol between the beer and the strip solution. Once the vapour pressure of water (less volatile than ethanol and other aroma compounds) is basically the same in both sides of the membrane, almost no water permeates to the strip solution.
Advantages:
- Dealcoholization is performed at low temperature
- The permeation of water is reduced
Disadvantages:
- Additional separation unit is required
- Need to recirculate the strip solution
- Loss of beer aroma compounds
- Dialysis is based on the use of membrane contactors (e.g., hollow-fibres) where beer flows counter-currently to the dialysate (e.g., water). Low molecular weight solutes, such as ethanol, cross the membrane as a result of the concentration gradient between two solutions. Ethanol permeates from beer to the dialysate until the equilibrium between concentrations at both sides of the membrane is reached. The resulted alcoholic stream can be sent to a distillation unit for ethanol removal and the aqueous stream reused in the dialysis unit to minimize the permeation of non-volatile compounds. Dialysis has a minimum impact in beer degradation since the separation process is carried out at atmospheric pressure and low temperature (below room temperature).
Advantages:
- Dealcoholization is performed at low temperature
- Water does not permeate the membrane
Disadvantages:
- Additional dialysis unit is required
- Need to recirculate the dialysate
- Loss of beer aroma compounds
- Pervaporation (PV) is a relative new membrane process that can be used for beer dealcoholization using hydrophobic membranes that favour the permeation of ethanol against the permeation of water. However, most of beer aroma compounds have higher affinity to hydrophobic membranes than ethanol, resulting in an aroma depleted dealcoholized beer. In this case, water is more permeable than ethanol. Thus, using a low pressure sweep stream of water vapour on the permeate side for keeping the partial pressure of all other beer compounds very low, the driving force of water is reduced and only ethanol and few aroma compounds permeate the membrane.
Advantages:
- Dealcoholization is performed at low temperature
- Ethanol removal is increased with hydrophilic membranes
- Water extraction is reduced by using a sweep gas with steam
Disadvantages:
- Additional pervaporation unit is required
- Aromas compound removal is higher than ethanol with hydrophilic membranes
- Permeation fluxes are low, demanding high membrane areas
- High costs of condensation and vacuum
Recovery of beer flavour compounds
The recovery of beer aroma compounds plays an important role during the production of non-alcoholic beer, since most of the above-mentioned processes for producing alcohol free beers result in a product whose aroma profile is unpleasant or distinct compared to the homologue alcoholic beer.
The recovery of natural beer compounds can be achieved by:
- Removing the lost aroma compounds, through side streams of the dealcoholization system;
- Extracting the aroma compounds directly from the original beer, before being submitted to dealcoholization Membrane processes seem to be the best approach to recover beer aroma compounds lost during ethanol removal. These separation processes have additional advantages when compared with traditional separation processes:
- Operation costs of membrane-based processes are usually low
- Can be carried out at low temperatures
- Do not need chemical additives, such as solvents or adsorbents
- Comparing the membrane processes such as nanofiltration, reverse osmosis, osmotic distillation and dialysis, show high permeability to ethanol compared to other aroma compounds, even employing membranes with low aroma compounds rejection. As a result, the permeate consists of an aqueous solution with high ethanol content. To improve the beer flavour of a dealcoholized beer, by returning the aroma extract to the dealcoholized beer, the resulting reconstructed beer will have an ethanol content above the legal limits or will display insufficient concentration of aroma compounds.
Pervaporation as a promising process for beer aroma recovery
As mentioned before, pervaporation (PV) is a membrane separation process based on cross-flow filtration, in which a feed liquid mixture contacts with a non-porous membrane and some of the feed compounds preferentially permeate through it. Vacuum is applied to the permeate side allowing the permeating species to leave the membrane in the vapour phase.
Nowadays, an increasing trend in the use of pervaporation for recovering aroma compounds in food applications, especially in the recovery and enrichment of aroma compounds from fruit juices as well as from fermented beverages, such as beer or wine.
Key beer aroma compounds
The most distinguishing aroma compounds are formed during beer maturation and are characteristic of a finished beer. These aroma compounds belong to two main chemical groups, higher alcohols and esters, and their concentration has a great impact on beer organoleptic quality.
The disadvantage of the dealcoholization processes is the loss of important aroma compounds. For overcoming the loss of aroma compounds, it was proposed to recover them by pervaporation before beer being submitted to dealcoholization and adding them to the dealcoholized beer. The pervaporation extraction of beer aroma compounds, such as higher alcohols and esters. The process proved to produce a very successful beer with a balanced aroma profile, similar to the original one.
Conclusions
In recent years, there has been an increased market share for no-alcohol and low alcohol beers. This is mainly due to the health reasons (such as obesity and other issues associated with high alcohol consumption), safety reasons (in the workplace or traffic roads) and increasingly strict social regulations and also the fact that alcohol consumption is forbidden in few countries because of religion as well as the awareness of problems that alcohol can bring about regarding civil responsibilities. No alcohol beers are recommended for specific groups of people such as pregnant women, sporting professionals, people with cardiovascular and hepatic pathologies, and medicated people. Consumers in such conditions are willing to have a beer as close to the conventional types as possible, from sensory point of view, a goal that is very difficult to achieve, because these beers suffer from having an artificial and immature flavour as well as inappropriate body and foam head. For these reasons, no-alcohol and low alcohol beverages have drawn social, technological, and economical interests. A variety of methods have been proposed and practiced for industrial production of no-alcohol and low alcohol beers with acceptable organoleptic properties.
Beer exerts various healthful effects on the human body due to its different constituents. However, consumption of no-alcohol beers does not provide the healthful effects of alcohol intake (in low amounts) as is the case with normal or low alcohol beers. On the other hand, it does not comprise adverse impacts of high alcohol intake caused by indiscriminate consumption of high-alcohol-containing beers. When consumed at moderate levels, low alcohol beers can be a good alternative to the regular alcoholic beers from the health point of view.













